Key takeaways:
- Clinical research suggests that CBD and other cannabinoids may help relieve MS symptoms such as muscle stiffness, pain, difficulty sleeping, and bladder problems.
- Most of the evidence is limited to studies using cannabis preparations with equal amounts of CBD and THC. More research on pure CBD and hemp-derived (high-CBD/low-THC) formulations is needed.
- While CBD is generally safe with minimal side effects, it may interact with some drugs used to treat MS. It’s best to talk to a doctor first if you’re taking any prescription medications.
More and more people are interested in using CBD to treat multiple sclerosis.
A survey from the MS Society UK found that 1 in 5 patients would consider using cannabis or cannabis-related products to help with their MS symptoms (1).
This makes sense because the usual treatments can have serious side effects whereas cannabis and CBD are seen as safer alternatives (2).
Although early studies suggest that CBD could be helpful, more high-quality clinical research is needed. Here’s what the science tells us about using CBD to help with multiple sclerosis.
Table of Contents
What Does the Research Say?
| Potential CBD Benefits | Strength of Evidence* |
| Reduced muscle spasms & stiffness | Medium |
| Reduced pain | Medium |
| Improved sleep | Medium |
| Improved bladder function | Low |
*Medium: several human studies with positive findings; Low: few human studies
Early studies suggest that CBD, THC, other cannabinoids, and whole-plant cannabis may have benefits for multiple sclerosis (MS).
However, most of the available research uses either whole-plant cannabis or Sativex, a 1:1 THC/CBD cannabis medicine, so it’s difficult to tell how much CBD contributes to the beneficial effects.
One of the most relevant studies looked at the effects of low-dose cannabis oil (CBD-rich, THC-rich, or 1:1) in 29 people with MS, finding that it reduced pain intensity, spasticity, and sleep problems while being safe and well-tolerated (3).
Muscle Spasticity
Muscle stiffness and spasms, referred to as spasticity, is a common MS symptom, with one survey suggesting that about 84% of patients suffer from it (4).
One 2019 systematic review of 14 studies found that 42-83% of MS patients taking Sativex experienced a 20% reduction in spasticity scores. The medication was well-tolerated without any significant side effects (5).
Another 2012 clinical trial evaluated the benefits of smoked cannabis for the management of muscle spasticity. Unlike placebo, cannabis was effective in managing the symptoms (6).
Pain Management
Pain is another common debilitating MS symptom that CBD might help with. A 2016 study evaluated the impact of Sativex on pain management in MS patients, reporting a significant reduction in neuropathic pain (7).
Meanwhile, a 2012 trial with 303 patients with MS-related neuropathic pain reported that Sativex was superior to placebo in providing pain relief (8). A similar 2018 study of Sativex reported significant reductions in pain in 19 MS patients (9).
Sleep Improvement
There is also evidence that cannabinoids may help with sleep; it’s estimated that half of patients with multiple sclerosis experience insomnia (10).
A 2007 review of Sativex for sleep found that it had noticeable benefits for patients with MS and other chronic pain conditions, with 40-50% of participants achieving good or very good sleep quality (11).
Bladder Function
MS patients frequently experience bladder dysfunction, which CBD might also help with. One 2017 study in MS patients found that Sativex resulted in fewer “overactive bladder” symptoms (12).
In summary, cannabinoids like CBD and THC hold real potential in relieving MS symptoms. The most recent 2023 review which analyzed 25 clinical studies with 3763 participants concluded that: (13)
- Cannabinoids significantly reduce muscle spasticity
- Show some improvement in chronic pain
- Improve the patient’s impression of their health
But it also called for further research and reported that a large number of people discontinued treatment due to side effects and an increased risk of nervous and psychiatric disorders, likely from the THC.
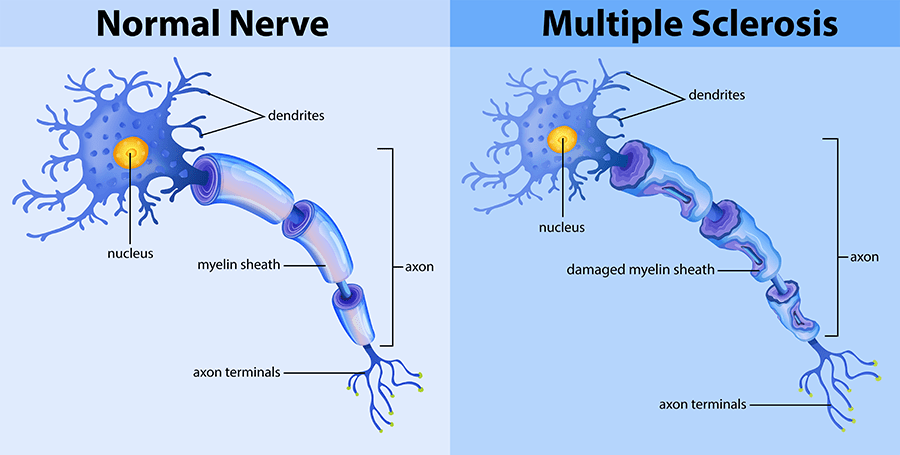
How Does CBD Help With MS?
CBD is just one of the several active compounds present in the cannabis plant. It might help manage the symptoms of multiple sclerosis through multiple ways.
CBD & the Endocannabinoid System
Scientists are still figuring out precisely how CBD works, but one way it seems to help is by interacting with the endocannabinoid system (ECS).
A complex signaling network that plays an important role in maintaining homeostasis (a state of healthy balance), the endocannabinoid system regulates key functions including sleep, appetite, emotion, metabolism, immunity, pain, cognition, and digestion (14).
It consists of endocannabinoids (cannabinoids made by our bodies) which interact with CB1 and CB2 receptors like a key that matches a lock, and enzymes that build and break down endocannabinoids.
One way CBD interacts with our ECS is by increasing the levels of anandamide, one of the two main endocannabinoids (15). Anandamide is known to suppress pain and have other beneficial effects (16).
CBD & Other Bodily Systems
Another way CBD works is by interacting with non-cannabinoid receptors such as TRPV1 and TRPV2. TRPV1 seems to have a role in sending messages to the body about pain. CBD inhibits the TRPV1 receptor which might help in pain relief (17).
There’s also growing evidence that CBD can help with MS by relieving inflammation and supressing the immune system because multiple sclerosis is believed to be an autoimmune disorder (18, 19).
How to Best Use CBD For MS?
CBD is commercially available in a variety of forms including oils, capsules, gummies, vapes, and topical products. Each form of CBD has its pros and cons.
Generally speaking, CBD oil is your best bet because it has high bioavailability (absorption), long-lasting effects, is easy to dose up or down, and is cost-effective.
You can also vape CBD if you want the fastest effects but vaping does come with certain health risks and also wears off faster than other methods — after about 1-4 hours (20).
Another option is gummies and capsules. However they have one key downside — because of first-pass metabolism in the digestive tract and liver, only a small amount (6-24%) of the CBD is absorbed and has a beneficial effect (21).
Finally, topical CBD products can be helpful for localized relief of MS spasticity or pain but are not ideal for body-wide symptoms.
Whatever option (or combination) you choose, we recommend going for full-spectrum CBD formulations because they may offer greater benefits from the synergy between their many beneficial components (the entourage effect) (22).

Dosage
At present, there is no officially recommended dose of CBD for the treatment of multiple sclerosis. The right dose depends on individual factors such as body weight, gender, genetics, and the type of product you’re using, so a personalized approach is necessary.
It’s best to go with the “start low and go slow” approach — start with a small amount like 10 mg and gradually increase the dose until you feel the desired effects (23).
Side Effects
CBD is considered a generally safe substance. According to a 2020 systematic review, CBD was well-tolerated with mild side effects in most clinical trials (24). Some potential side effects associated with CBD include (25):
- Drowsiness
- Fatigue
- Dry mouth
- Diarrhea
- Decrease in appetite
A 2021 safety study of low-dose cannabis oils in multiple sclerosis also found that they were generally safe and well-tolerated (3).
Drug Interactions
CBD may potentially interact with over-the-counter and prescription drugs, including those used for MS treatment (26).
Since patients with multiple sclerosis might be taking several prescription medications, there is a risk of drug interactions so it’s important to talk to your doctor before taking CBD.
CBD & MS: Promising, But More Research Needed
CBD and cannabis as a whole are receiving growing attention from both researchers and MS sufferers because current treatment options can be ineffective or cause significant side effects.
Although more research is needed — particularly on pure CBD and hemp-derived, full-spectrum extracts — current evidence suggests that cannabinoids may help with the symptoms of MS.
If you’re considering giving CBD a try, we recommend trying a high-quality full-spectrum CBD oil and gradually raising the dosage until you start to experience the desired relief.
References
- https://www.mssociety.org.uk/about-ms/treatments-and-therapies/cannabis
- Kołtuniuk, Aleksandra, and Justyna Chojdak-Łukasiewicz. “Adherence to Therapy in Patients with Multiple Sclerosis.” International Journal of Environmental Research and Public Health 19.4 (2022): 2203.
- Gustavsen, S., et al. “Safety and efficacy of low-dose medical cannabis oils in multiple sclerosis.” Multiple Sclerosis and Related Disorders 48 (2021): 102708.
- Fernández, Óscar, et al. “The broad concept of “Spasticity-Plus Syndrome” in multiple sclerosis: a possible new concept in the management of multiple sclerosis symptoms.” Frontiers in Neurology 11 (2020): 152.
- Akgün, Katja, et al. “Daily practice managing resistant multiple sclerosis spasticity with delta-9-tetrahydrocannabinol: cannabidiol oromucosal spray: a systematic review of observational studies.” Journal of central nervous system disease 11 (2019): 1179573519831997.
- Corey-Bloom, Jody, et al. “Smoked cannabis for spasticity in multiple sclerosis: a randomized, placebo-controlled trial.” Cmaj 184.10 (2012): 1143-1150.
- Russo, Margherita, et al. “Evaluating Sativex® in neuropathic pain management: a clinical and neurophysiological assessment in multiple sclerosis.” Pain Medicine 17.6 (2016): 1145-1154.
- Langford, R. M., et al. “A double-blind, randomized, placebo-controlled, parallel-group study of THC/CBD oromucosal spray in combination with the existing treatment regimen, in the relief of central neuropathic pain in patients with multiple sclerosis.” Journal of neurology 260 (2013): 984-997.
- Turri, Mara, et al. “Pain modulation after oromucosal cannabinoid spray (SATIVEX®) in patients with multiple sclerosis: a study with quantitative sensory testing and laser-evoked potentials.” Medicines 5.3 (2018): 59.
- Alhazzani, A. A., et al. “Insomnia among non-depressed multiple sclerosis patients: a cross-sectional study.” The Egyptian journal of neurology, psychiatry and neurosurgery 54 (2018): 1-5.
- Russo, Ethan B., Geoffrey W. Guy, and Philip J. Robson. “Cannabis, pain, and sleep: lessons from therapeutic clinical trials of Sativex®, a cannabis‐based medicine.” Chemistry & biodiversity 4.8 (2007): 1729-1743.
- Maniscalco, Giorgia Teresa, et al. “THC/CBD oromucosal spray in patients with multiple sclerosis overactive bladder: a pilot prospective study.” Neurological Sciences 39 (2018): 97-102.
- Amatya, Bhasker, and Fary Khan. “Are Cannabis and Cannabinoids Effective for Symptomatic Treatment in People With Multiple Sclerosis?: A Cochrane Review Summary With Commentary.” American Journal of Physical Medicine & Rehabilitation 102.1 (2023): 83-84.
- Komarnytsky, Slavko, et al. “Endocannabinoid system and its regulation by polyunsaturated fatty acids and full spectrum hemp oils.” International Journal of Molecular Sciences 22.11 (2021): 5479.
- de Almeida, Douglas L., and Lakshmi A. Devi. “Diversity of molecular targets and signaling pathways for CBD.” Pharmacology research & perspectives 8.6 (2020): e00682.
- Clapper, Jason R., et al. “Anandamide suppresses pain initiation through a peripheral endocannabinoid mechanism.” Nature neuroscience 13.10 (2010): 1265-1270.
- Anand, Uma, et al. “CBD effects on TRPV1 signaling pathways in cultured DRG neurons.” Journal of Pain Research (2020): 2269-2278.
- Al-Ghezi, Zinah Zamil, et al. “Combination of cannabinoids, Δ9-tetrahydrocannabinol and cannabidiol, ameliorates experimental multiple sclerosis by suppressing neuroinflammation through regulation of miRNA-mediated signaling pathways.” Frontiers in immunology 10 (2019): 1921.
- Furgiuele, Alessia, et al. “Immunomodulatory potential of cannabidiol in multiple sclerosis: A systematic review.” Journal of Neuroimmune Pharmacology 16 (2021): 251-269.
- Borodovsky, Jacob T., et al. “Smoking, vaping, eating: Is legalization impacting the way people use cannabis?.” International Journal of Drug Policy 36 (2016): 141-147.
- Perucca, Emilio, and Meir Bialer. “Critical aspects affecting cannabidiol oral bioavailability and metabolic elimination, and related clinical implications.” CNS drugs 34 (2020): 795-800.
- Russo, Ethan B. “The case for the entourage effect and conventional breeding of clinical cannabis: no “strain,” no gain.” Frontiers in plant science (2019): 1969.
- Lucas, Catherine J., Peter Galettis, and Jennifer Schneider. “The pharmacokinetics and the pharmacodynamics of cannabinoids.” British journal of clinical pharmacology 84.11 (2018): 2477-2482.
- Larsen, Christian, and Jorida Shahinas. “Dosage, efficacy and safety of cannabidiol administration in adults: a systematic review of human trials.” Journal of clinical medicine research 12.3 (2020): 129.
- https://www.cdc.gov/marijuana/featured-topics/CBD.html
- Balachandran, Premalatha, Mahmoud Elsohly, and Kevin P. Hill. “Cannabidiol interactions with medications, illicit substances, and alcohol: a comprehensive review.” Journal of general internal medicine 36 (2021): 2074-2084.

Gleb is a freelance writer from Vancouver, Canada specializing in CBD and cannabis. He’s read thousands of studies on CBD and other supplements, helping him translate complex science into plain language. Gleb has tried and reviewed dozens of CBD brands and products, written third-party testing reports, and knows the CBD industry inside and out. When not writing, he likes to kickbox, travel, and tell everyone how awesome intermittent fasting is.